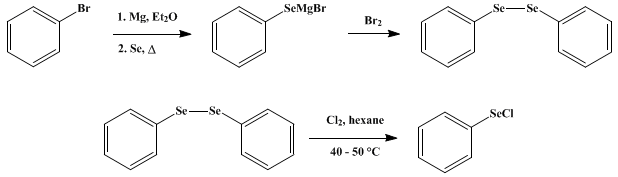
A Benzoyl Peroxide/Diphenyl Diselenide Binary System for Functionalization of Alkynes Leading to Alkenyl and Alkynyl Selenides. | Semantic Scholar

Nanomolar Detection of Palladium (II) through a Novel Seleno-Rhodamine-based fluorescent and colorimetric chemosensor - ScienceDirect

Synthesis of selenol esters: palladium-catalyzed coupling of phenyl tributylstannyl selenide with aryl iodides and carbon monoxide - ScienceDirect

Palladium-catalyzed alkynylselenation of acetylenedicarboxylates leading to enyne selenides and application to synthesis of multisubstituted aryl selenides - ScienceDirect
![Copper/Palladium‐Catalyzed Cyclization/Cross‐Coupling Cascade Reaction of 2‐gem‐Dibromovinyl Aryl Selenides: Synthesis of 2‐Substituted Benzo[b]selenophenes - Bilheri - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library Copper/Palladium‐Catalyzed Cyclization/Cross‐Coupling Cascade Reaction of 2‐gem‐Dibromovinyl Aryl Selenides: Synthesis of 2‐Substituted Benzo[b]selenophenes - Bilheri - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/3b49103f-96fe-406e-a2c3-93fe446e24f8/adsc201701095-toc-0001-m.jpg)
Copper/Palladium‐Catalyzed Cyclization/Cross‐Coupling Cascade Reaction of 2‐gem‐Dibromovinyl Aryl Selenides: Synthesis of 2‐Substituted Benzo[b]selenophenes - Bilheri - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library

Palladium Complex Immobilized on Magnetic Nanoparticles Modified with 2-Aminopyridine Ligand: A Novel and Efficient Recoverable Nanocatalyst for C–S and C–Se Coupling Reactions | SpringerLink

Rational Synthesis of Palladium Nanoparticles Modified by Phosphorous for the Conversion of Diphenyl Ether to KA Oil - ScienceDirect

Palladium‐Catalyzed Picolinamide‐Directed Benzylic C(sp3)−H Chalcogenation with Diaryl Disulfides and Diphenyl Diselenide - Wang - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

A Benzoyl Peroxide/Diphenyl Diselenide Binary System for Functionalization of Alkynes Leading to Alkenyl and Alkynyl Selenides. | Semantic Scholar

Functional Polyselenoureas for Selective Gold Recovery Prepared from Catalyst-Free Multicomponent Polymerizations of Elemental Selenium | CCS Chem

Homoleptic Platinum(II) and Palladium(II) Organothiolates and Phenylselenolates: Solvothermal Synthesis, Structural Determination, Optical Properties, and Single‐Source Precursors for PdSe and PdS Nanocrystals - Chui - 2010 - Chemistry – An Asian ...

Synthesis of silica-supported selenide palladium(0) complex and its catalytic properties for phenylation of acid chlorides and aryl iodides or bromides - ScienceDirect

A Benzoyl Peroxide/Diphenyl Diselenide Binary System for Functionalization of Alkynes Leading to Alkenyl and Alkynyl Selenides. | Semantic Scholar
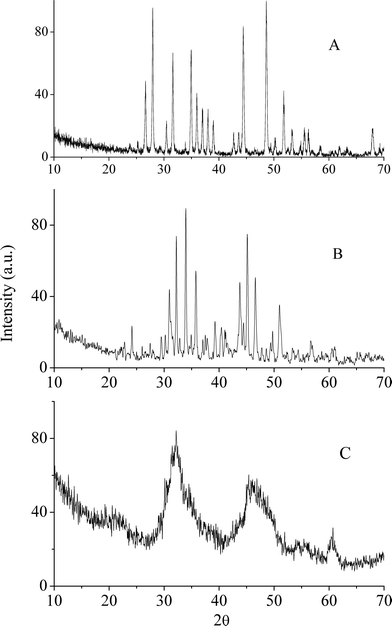
Oligomeric allyl-palladium( ii ) complexes of β-substituted ethylselenolates: Syntheses, structures and thermal decomposition - Dalton Transactions (RSC Publishing) DOI:10.1039/C1DT10392J

Modular chiral selenium-containing oxazolines: synthesis and application in the palladium-catalyzed asymmetric allylic alkylation - ScienceDirect
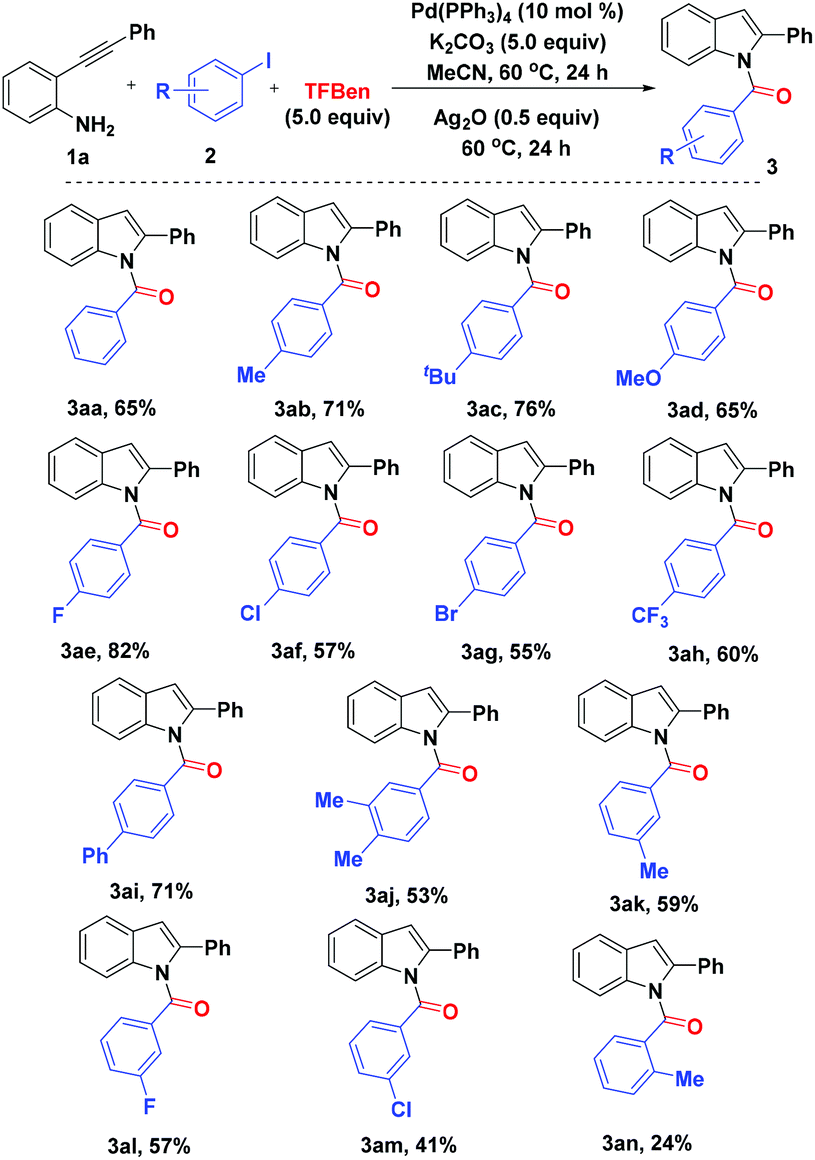
Palladium-catalyzed carbonylative cyclization of 2-alkynylanilines and aryl iodides to access N-acyl indoles - Organic Chemistry Frontiers (RSC Publishing)

Palladium‐Catalyzed Picolinamide‐Directed Benzylic C(sp3)−H Chalcogenation with Diaryl Disulfides and Diphenyl Diselenide - Wang - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library


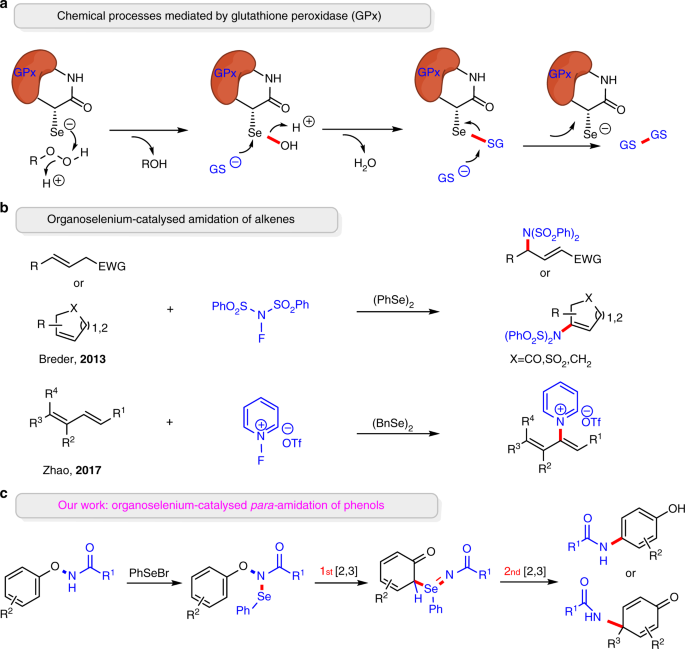

/48A57479F8045CEA802585F9008252CA/$file/FT10775_structure.png)